Research
Synthetic Methodology involving the generation of the 1,1,2,2-Tetrafluoroethyl Radical
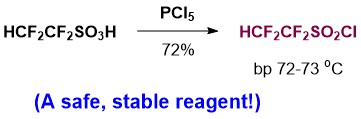
Synthesis of 1,1,2,2-Tetrafluoroethanesulfonylchloride and its Use For Introduction of the 1,1,2,2-Tetrafluoroethyl Group Into Organic Compounds
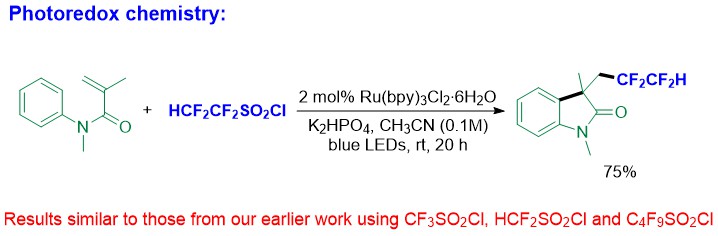
1,1,2,2-Tetrafluoroethyl-containing compounds are valuable structures due to their unique physicochemical properties, which have increasing potential application in drug discovery. However, synthetic methods for preparing such compounds are rare. We have developed a method to prepare 1,1,2,2-tetrafluoroethanesulfonyl chloride as well as methods for its use to introduce the HCF2CF2 group into organic molecules photoinduced and photoredox chemistry.


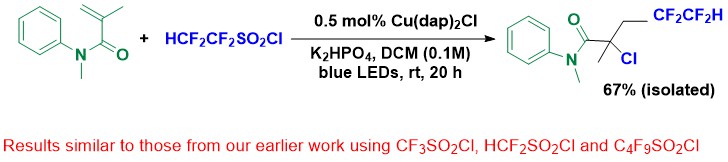
In contrast, there is the photo-induced atom transfer reaction with Cu catalysis:
The use of 1,1,2,2-Tetrafluoroethanesulfinate to introduce the HCF2CF2 group into organic compounds

The Pentafluorosulfanyl (SF5) group.
Since early in 2000’s, our group has played a leadership role in the development of methodology for incorporation of the SF5 group into aliphatic and aromatic compounds. Until then, in spite of its unique steric and electronic properties, it had been largely ignored by pharmaceutical and agrochemical companies. No longer.

Novel Synthetic Chemistry of Derivatives of Octafluoro[2.2]paracyclophane
Since the first isolation of the parent compound in 1949, interest in molecules containing the unique structural features of [2.2]paracyclophanes has never waned and has generated a literature filled with unusual structural features and chemistry. This chemistry includes their unique role as CVD precursors of what are known as Parylene polymers.
Our research group has been involved in studies of octafluoro-[2.2]-paracyclophane since 1990, work including development of three synthetic methods for the compound, which has considerable commercial interest as precursors to its parylene polymer, which has exceptional thermal stability and dielectric properties.
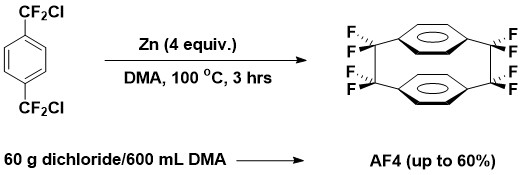
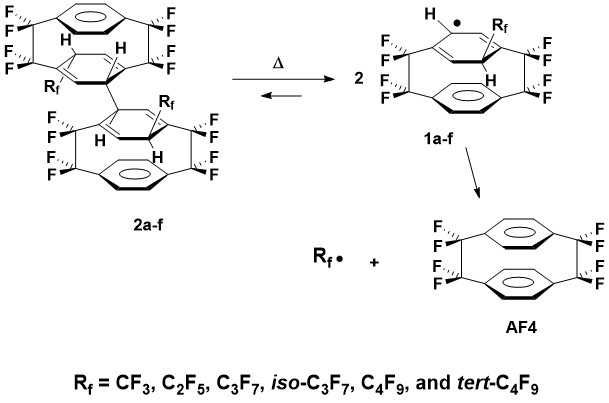
Our current research revolves around building on earlier published results to develop AF4-based compounds that will serve as clean fluorinated radical sources: